
The literature generally describes the chromophores of vision as made up of a protein combined with a retinal. This view is simplistic. The chromophores of vision consist of modified molecules derived from Vitamin A by oxygenation. These chromophores are then deposited on a protein substrate (opsin) that forms the disks of the outer segment of each photoreceptor neuron. The following figure shows the four chromophores of vision used by humans, and other saline based animals. A slightly different ring structure is used in fresh-water fish (Vitamin A2) and in certain flies who eat decomposing matter (Vitamin A3). Click here to review these Vitamin A's
RESONANT MOLECULES-- 2 OXYGEN'S
MEMBERS OF THE "INDICATOR" FAMILY
DERIVATIVES OF RETINOL
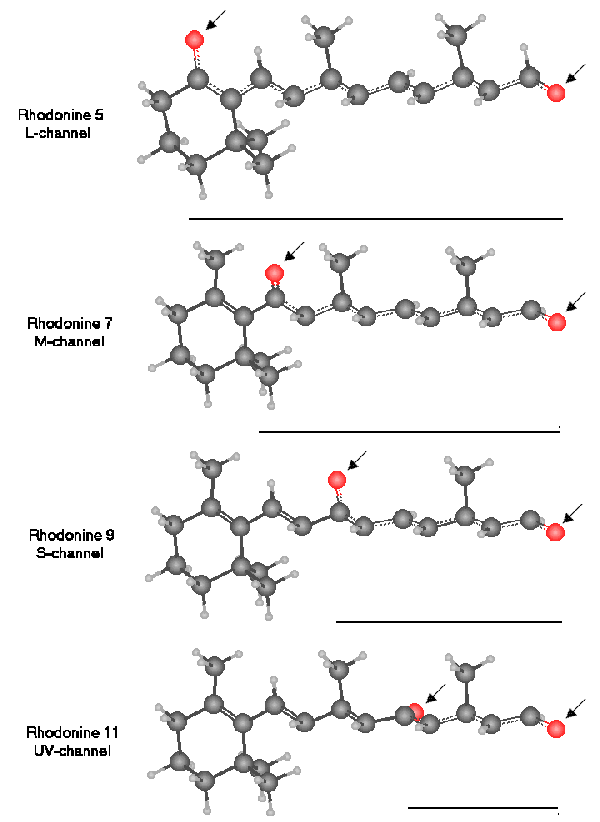
The STRUCTURE OF THE RHODONINES [from Section 5.5.9]
All of the molecules have similar molecular weights. All exhibit a peak absorption in dilute solution near 493 nm. Only when in liquid crystalline state do they exhibit their biologically significant anisotropic spectrums.
Note the resonant structure between the two oxygens is separate from, but shares much of the same carbon chain with, the conjugate structure of the parent retinoid.
These spectrums peak at 625 nm for Rhodonine(5), 532 nm for Rhodonine (7), 437 nm for Rhodonine (9) and 342 nm for Rhodonine (11).
The bars under the molecules indicate the resonant length of each molecule on a relative scale. These lengths are proportional to the wavelengths of the resonant peaks of the molecules.
Return to the website home page